 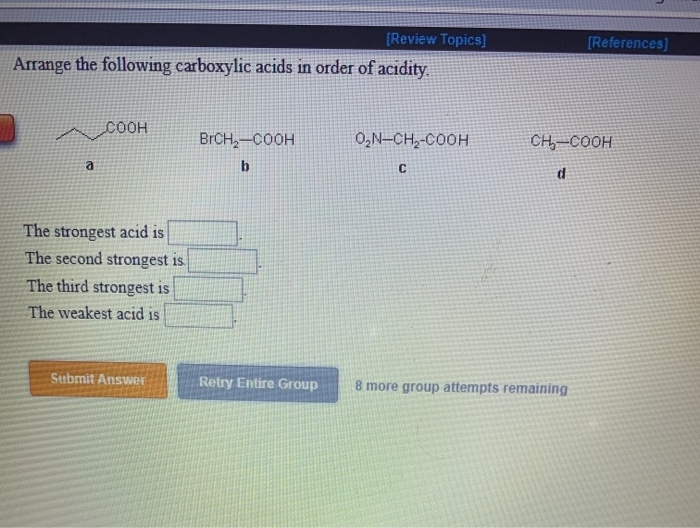
Um, the un substituted for I guess the the aromatic. The pK a s of some typical carboxylic acids are listed in the following table. 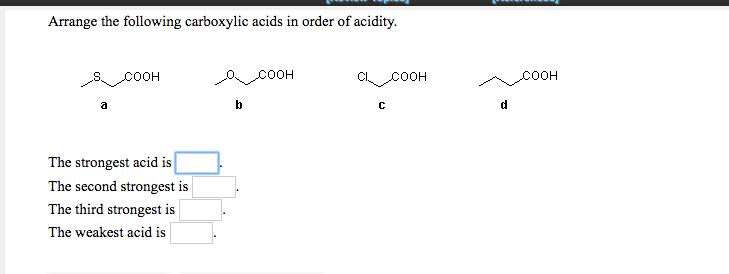
It acts as an electron donating group, and so decreases the acidity of the carb. Which carboxylic acid gives Volhard Zelinsky reaction In this article we introduce the Hell-Volhard-Zelinsky reaction of carboxylic acids, which results in an alpha bromo carboxylic acid.After summarizing what the reaction does, well go through the four stages of the mechanism that eventually result in the formation of a new C-Br bond with the breakage of a C-H bond. So, um, are least acidic is going to be, uh, this third molecule with the metal group. If you have any queries, drop a comment below and we will get back to you. In order to make sense of this trend, we will once again consider the stability of the. Transcribed image text: Arrange the following carboxylic acids in order of acidity: Indicate, by letter, the strongest acid and the weakest acid of those. Oxalic acid and electron withdrawing groups are going to increase the acidity of the car back. We hope this article on ‘Acidity of Carboxylic Acids’ has helped you. Which is more acidic, alcohol or phenol?Īns: Phenol is more acidic than alcohol since phenoxide ion is more stabilized than alkoxide ion due to the resonance effect. Hydrogen and metals combined in an alkaline solution produce. Therefore, carboxylic acids are stronger acids than alcohol. Carbaryl acid is the name given to organic compounds that contain carboxylic acid groups. However, in alcohols, no resonance is possible for the alcohols as well as alkoxide ions. The carboxylic acids react with metals to liberate hydrogen and are soluble in both NaOH and NaHCO 3 solutions. Carbon compounds that contain carboxyl group \(\left( \) forming stable carboxylate ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
